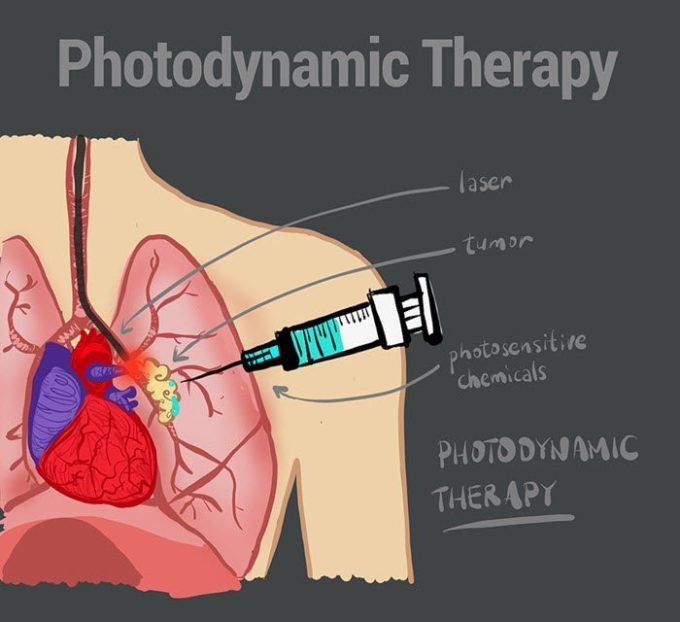
Treatment for the asbestos cancer mesothelioma is classified as one of two types — curative or palliative.
- Curative therapy focuses on killing the cancer cells and stopping further growth.
- Palliative care treatments aim at relieving the symptoms and improving the patient’s quality of life.
Regardless of the approach taken, the primary doctor will closely monitor the patient’s response to therapy and make any needed adjustments to the treatment plan.
In many instances, a patient will begin treatment with an aggressive protocol, but will be transitioned to a more palliative treatment plan if the disease stops responding to the aggressive therapies.
An empowered mesothelioma patient is often the best patient for a medical team. Empowered patients are informed of their disease’s track and treatment options, and they are willing to take an active role in their treatment strategies.
Knowing what questions to ask of your physician is an important step toward becoming an empowered mesothelioma patient.
Learning about the benefits and risks of each treatment option is critical to ensuring you are prepared for the arduous battle ahead. Listen closely to your doctor, and ask as many questions as you need to get the full picture.