Mesothelioma Clinical Trials

When you and your doctor developed a mesothelioma treatment plan and talked about your long term care options, you more than likely discussed the possibility of one day participating in a clinical trial. If your cancer is returning in spite of surgery, chemotherapy, and radiation, the time to start considering a clinical trial might be now.
A clinical trial gives people an experimental disease treatment developed in a laboratory to see whether that treatment is safe and effective for patients. Trials are tightly controlled and highly professional, but there is still a risk of patient harm and good results are not guaranteed.
On the upside, trials may produce major medical breakthroughs that permanently alter the treatment paradigm for a disease. Many of the treatments of tomorrow are only available through clinical trials. Still, you shouldn’t participate in a trial before you’ve tried more tried and true standard treatments. Those treatments, after all, only became standard after they were tested in clinical trials.
Finding a Clinical Trial Research Program
Deciding you want to take part in a clinical trial does not guarantee your place in one. Trials are limited and have very specific eligibility criteria.
The first thing you’ll need to do is find a trial that seems like a good fit.
Then, you’ll need to be medically evaluated for patient requirements such as age, disease stage, ethnicity, gender, and medical history.
Here are some resources for finding a mesothelioma clinical trial:
Your Doctor
The first person to ask which clinical trial you should participate in is your doctor. If he or she is at a mesothelioma specialty center, there is probably a trial being run by the center or an affiliated university. If there are no trials nearby, your doctor has the resources and connections to either find one for you or to direct you where to look.
The Mesothelioma Applied Research Foundation.
Mary Hesdorffer is the Executive Director at the Meso Foundation and is also a Nurse Practitioner who spends some of her time working at the National Cancer Institute. Mary has expertise in the development and implementation of clinical trials, and she was instrumental in the development of the Meso Foundation’s book, “Mesothelioma Clinical Trials.” Mary will not hesitate to talk to anyone about mesothelioma and clinical trials and is available by phone or email. When talking to mesothelioma patients, Mary is able to offer them sound, unbiased advice.
Visit curemeso.org for more information.
Friends, Family & Other Mesothelioma Patients
Sometimes there is nothing more effective than getting information through the grapevine. It’s surprising how quickly you will find out who knows what or whom when the word gets out that you or a family member has mesothelioma.
Don’t be shy about taking the initiative and asking people—it could make a difference in your life. Also, check with your doctor’s office or local medical center for names of mesothelioma patients who have offered to talk with others. Other good places to ask around is in your place of worship, senior center or other local gathering places.
Online
The researchers running clinical trials are required to maintain the information through a national registry. For a complete listing of available trials go to ClinicalTrials.gov and search for ‘mesothelioma.’ The National Cancer Institute also maintains a database of more than 12,000 clinical trials accepting participants, as well as more than 25,000 clinical trials that are no longer recruiting.
Social Media
Join in the many mesothelioma communities on Facebook to find out about clinical trials patients are participating in and newly announced trials. There are many groups out there, and the members are extremely supportive. Mavis Nye of the UK, a six-year mesothelioma survivor, is one of the most well-known patient advocates and an administrator of one of the largest mesothelioma communities on Facebook.
Contact Mavis for more information.
https://www.facebook.com/mavis.nye
Trial guidelines
US clinical trials are closely monitored and run according to strict guidelines. The National Institutes of Health explains that federally funded clinical mesothelioma research has safeguards in place to protect the participants, including explaining to the participants:
- What will happen during a study including eligibility criteria, evaluations and all procedures to be performed;
- The possible risks, benefits, alternatives, and responsibilities of the clinical trial are explained thoroughly to patients before they agree to participate;
- That a clinical study is reviewed by an independent review board prior to the start to ensure that potential risks are clearly stated, and that investigators take sufficient and appropriate measures to address risks before the study proceeds;
- The study will be closely monitored once it is underway by an independent committee for safety and conformation to the study documents;
- That their privacy will be protected; and
- The study will be ethical and will follow international and federal “good clinical practices” guidelines.
How Clinical Trials Work
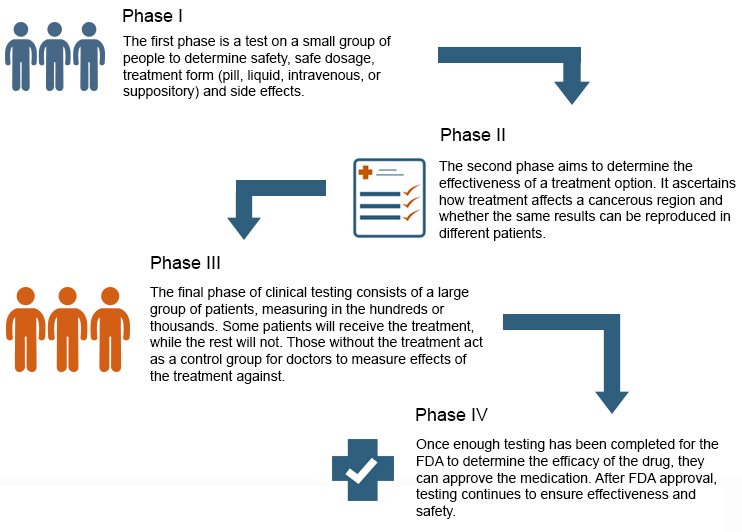
Before a new treatment is approved, it usually goes through the three phases described below:
Why You Should Participate In Mesothelioma Research Programs
 New Treatment Options
New Treatment Options
Patients can have access to innovative treatments before they are available to everyone. Every new treatment has started out as a clinical trial.
 The possibility of a Cure
The possibility of a Cure
Since this is a new treatment, there is a possibility that it can save your life.
Learn More About Clinical Trials
The best thing you can do to better understand what a clinical trial entails is to talk to a patient who has participated in one, or a family member who was there every step of the way. While the medical team can answer the critical health-related questions, a participant is the only person who can tell you the impact from the family’s and patient’s perspective.
Not everyone, though, has the luxury of knowing such a person. That’s why MesotheliomaHelp has a free resource, created with help from our Faces of Mesothelioma Community, on what to expect when participating in a clinical trial.
We’ve also put together this SlideShare presentation, which explains some of the science and history behind clinical trials.
Open Studies
Below is a partial list of active clinical trials for mesothelioma. For a complete list see www.clinicaltrials.gov.
https://www.clinicaltrials.gov/
Study 1: Do Your Genes Put You at a Higher Risk of Developing Mesothelioma
https://clinicaltrials.gov/show/NCT01590472
Study 2: Intrapleural Administration of HSV1716 to Treat Patients With Malignant Pleural Mesothelioma.
https://clinicaltrials.gov/show/NCT01721018
Study 3: Tissue Procurement and Natural History Study of Patients With Malignant Mesothelioma
https://clinicaltrials.gov/show/NCT01950572
Study 4: SS1P and Pentostatin Plus Cyclophosphamide for Mesothelioma
https://clinicaltrials.gov/show/NCT01362790
Study 5: Placebo Controlled Study of VS-6063 in Subjects With Malignant Pleural Mesothelioma
https://clinicaltrials.gov/show/NCT01870609
Study 6: Alisertib in Malignant Mesothelioma
https://clinicaltrials.gov/show/NCT02293005
Study 7: Vinorelbine in Mesothelioma
https://clinicaltrials.gov/show/NCT02139904
Study 8: Pemetrexed Disodium or Observation in Treating Patients With Malignant Pleural Mesothelioma Without Progressive Disease After First-Line Chemotherapy
https://clinicaltrials.gov/show/NCT01085630
Study 9: Safety and Efficacy of Oshadi D and Oshadi R for Malignant Mesothelioma Treatment
https://clinicaltrials.gov/show/NCT01627795
Study 10: PIT: Prophylactic Irradiation of Tracts in Patients With Malignant Pleural Mesothelioma
https://clinicaltrials.gov/show/NCT01604005
Sources:
- National Institutes of Health
http://clinicalcenter.nih.gov/participate/faqaboutcs.shtml - ClinicalTrials.gov
https://clinicaltrials.gov/ - National Cancer Institute
https://www.cancer.gov/about-cancer/treatment/clinical-trials/search
 New Treatment Options
New Treatment Options The possibility of a Cure
The possibility of a Cure