American Society of Clinical Oncology (ASCO) releases update to its pleural mesothelioma treatment guidelines

Earlier this year, the American Society of Clinical Oncology (ASCO) published an update to its 2018 guideline for the treatment of pleural mesothelioma. This summary of the guidelines is based on the published manuscript by Kindler HL, Dagogo-Jack I, de Perrot M, Drazer MW, Ismaila N, Hassan R. Treatment of Pleural Mesothelioma: ASCO Guideline Clinical Insights. JCO Oncol Pract. 2025 Mar 7:OP2500035. doi: 10.1200/OP-25-00035. Epub ahead of print. PMID: 40053896.
To update this document, mesothelioma experts from fields of medical oncology, surgery, pathology, radiation oncology and others, convened to review and analyze 110 relevant studies for each recommendation presented. The recommendations were divided into categories such as diagnosis, surgery, immunotherapy, chemotherapy, radiation therapy, pathology, and germline testing.
Researchers and providers who contributed to these treatment guidelines are affiliated with the following centers:
- University of Chicago Medicine
- American Society of Clinical Oncology
- University of California San Diego Moores Cancer Center
- Cross Cancer Institute, University of Alberta
- University of Wisconsin-Madison and Carbone Cancer Center
- Massachusetts General Hospital Cancer Center
- Johns Hopkins University
- Memorial Sloan Kettering Cancer Center
- Princess Margaret Cancer Centre – Toronto, Canada
- Center for Cancer Research, National Cancer Institute
- Levine Cancer Institute
- National Cancer Institute, Egypt
- Mesothelioma Applied Research Foundation
DIAGNOSIS AND STAGING
Mesothelioma patients often present with certain generalized symptoms that don’t go away after treatment. Pleural effusions, an uncomfortable accumulation of fluid in the pleural space that makes it difficult to expand the lung and breathe, are one of the most common symptoms.
Initial Assessment: In patients presenting with fluid, thoracentesis for pleural effusions, followed by cytologic examination is strongly recommended.
Biopsy: Thoracoscopic biopsy is strongly recommended when treatment is planned; use minimal incisions in potential surgical fields.
Alternative Biopsy Methods: Use core needle or open pleural biopsy only when thoracoscopic biopsy is not feasible.
Imaging:
- CT chest/abdomen with contrast and FDG PET/ CT for staging. (FDG is radioactive glucose injected into the bloodstream to see which areas of the tumor are metabolically active and how active they are.
- MRI (optional) to evaluate chest wall / diaphragm / mediastinum invasion.
Invasive Staging: Mediastinoscopy and/or EBUS for PET-avid nodes in candidates for maximal surgical cytoreduction; laparoscopy for suspected abdominal spread.
Measurement & Staging:
- Expert radiologist should perform measurements on CT scan based on modified RECIST 1.1 criteria.
- Follow mRECIST 1.1 for response evaluation; CT-based tumor volume measurement remains investigational.
SURGERY
Mesothelioma surgery has been at the center of debate since the release of the United Kingdom’s phase III study results from the Mesothelioma and Radical Surgery (MARS) 2 randomized controlled clinical trial. This was the first such trial in mesothelioma. The study results showed that patients treated with chemotherapy alone fared better than those who underwent surgery. However, researchers caution that interpretation of the study ought to consider data from nonrandomized surgical studies which show better outcome and much lower 30 and 90-day mortality. As a result, surgery should still be considered for patients with favorable prognostic characteristics.
General Guidance: Surgery should not be based on resectability alone. It’s reserved for select patients with early-stage (T1-3N0), epithelioid histology.
Surgical Options:
- Lung-sparing procedures (PD/EPD) preferred over EPP.
- EPP reserved for exceptional cases at high-volume centers.
Multimodality Care: Surgery should be part of a multidisciplinary approach including chemo/immunotherapy.
Neoadjuvant or Adjuvant Therapies:
- Chemotherapy (pemetrexed/platinum) may be given pre- or postoperatively.
- Neoadjuvant immunotherapy is an option; adjuvant immunotherapy is not recommended due to limited data currently available.
Palliative Surgery: For patients unfit for full cytoreduction, procedures like tunneled catheters or pleurodesis are options.
Intracavitary Therapies: May be safe at experienced centers, but benefits are unclear.
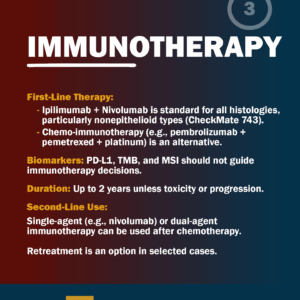
IMMUNOTHERAPY
First-Line Therapy:
- Ipilimumab + Nivolumab is standard for all histologies, particularly nonepithelioid types (CheckMate 743).
- Chemo-immunotherapy (e.g., pembrolizumab + pemetrexed + platinum) is an alternative.
Biomarkers: PD-L1, TMB, and MSI should not guide immunotherapy decisions.
Duration: Up to 2 years unless toxicity or progression.
Second-Line Use:
- Single-agent (e.g., nivolumab) or dual-agent immunotherapy can be used after chemotherapy.
- Retreatment is an option in selected cases.
CHEMOTHERAPY
Chemotherapy was for many years the only standard treatment approved for mesothelioma. This treatment regimen is most familiar to researchers and physicians alike ensuring the most consistent side effect control. However, chemotherapy for mesothelioma has many limitations. For instance, patients with the nonepithelioid subtype do not respond to chemotherapy very well.
First-Line:
- Epithelioid: Pemetrexed + platinum ± bevacizumab.
- Nonepithelioid: Prefer immunotherapy; use chemotherapy only if immunotherapy is contraindicated.
Maintenance: Pemetrexed maintenance not recommended; gemcitabine maintenance may be offered.
Second-Line:
- After immunotherapy: Use pemetrexed + platinum ± bevacizumab.
- After both: Options include gemcitabine ± ramucirumab or vinorelbine.
Special Populations:
- PS 2: Single-agent chemo may be offered.
- PS ≥3: Palliative care recommended.
Duration: 4–6 cycles recommended; break afterward if stable or responding.
RADIATION THERAPY
Radiation therapy for mesothelioma is typically used in conjunction with other treatments to reduce chances of local recurrence. Sometimes, it is also used to shrink tumors as symptom-management.
Prophylactic Radiation: Not recommended to prevent tract recurrence.
Adjuvant Use:
- May reduce local recurrence; recommend in experienced centers.
- Use hemithoracic IMRT post-EPP or post-PD/EPD (preferably in trials).
Palliative Use: Strongly recommended for symptom control using standard regimens.
Neoadjuvant Radiation: Due to potential for severe pulmonary toxicity, neoadjuvant radiation is not recommended with lung-sparing surgery.
Techniques: Use 2D, 3D, IMRT, or electrons depending on tumor site and organs at risk.
PATHOLOGY
The field of pathology is crucial in diagnosing the exact type of tumor in order to help determine the appropriate treatment.
Terminology: Non-malignant mesotheliomas have now been recategorized and renamed, so use of term “mesothelioma” without “malignant” preceding it, is now appropriate.
Diagnostic Tools: IHC with both positive and negative mesothelioma markers.
Subtype Reporting: Required: epithelioid, sarcomatoid, or biphasic, with detailed grading.
Biomarkers: Non-tissue-based biomarkers (e.g., serum tests) are not reliable for monitoring.
MIS (Mesothelioma In Situ): Consider in recurrent unilateral effusions; test with BAP1 and MTAP IHC.
Free Mesothelioma Patient & Treatment Guide
We’d like to offer you our in-depth guide, “A Patient’s Guide to Mesothelioma,” absolutely free of charge.
It contains a wealth of information and resources to help you better understand the condition, choose (and afford) appropriate treatment, and exercise your legal right to compensation.
Download Now